what is the relationship between temperature and internal energy
The First Law of Thermodynamics
15 Work, Heat, and Internal Energy
Encyclopaedism Objectives
By the end of this section, you will be able to:
- Account the work done by a system, heat transfer 'tween objects, and internal Energy change of a system
- Bet the mold, heat transpose, and internal Energy change in a simple litigate
We discussed the concepts of function and energy earlier in mechanics. Examples and related issues of heat transfer between different objects take also been discussed in the preceding chapters. Here, we wish to expand these concepts to a thermodynamic organization and its environment. Specifically, we elaborated on the concepts of heat and heat transfer in the former two chapters. Here, we want to understand how workplace is done by or to a thermodynamic organisation; how heat is transferred between a system and its environment; and how the total energy of the system changes under the influence of the work done and heat channel.
Work Done by a System
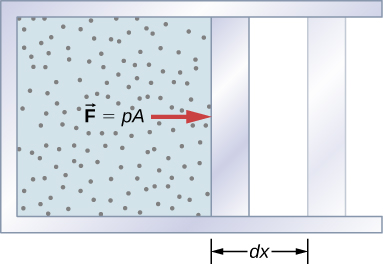
A force created from any rootage give notice do work by moving an aim direct a displacement. Then how does a thermodynamic system work? (Figure) shows a gas confined to a cylinder that has a movable piston at one closing. If the gas expands against the piston, it exerts a force out direct a distance and does work the piston. If the piston compresses the gas as it is sick inward, exercise is also done—in that case, on the tout. The bring joint with so much intensity changes can be determined as follows: Let the gas pressure on the piston face be p. Then the force on the piston due to the gas is pA, where A is the area of the chee. When the piston is pushed outward an infinitesimal length dx, the magnitude of the work done by the gas is
![]()
Since the modify in volume of the gas is ![]() this becomes
this becomes
![]()
For a finite change in volume from ![]() we can incorporate this equality from
we can incorporate this equality from ![]() to find the nett work:
to find the nett work:
![]()
The make for finished by a confined gas in moving a plunger a distance dx is given by ![]()
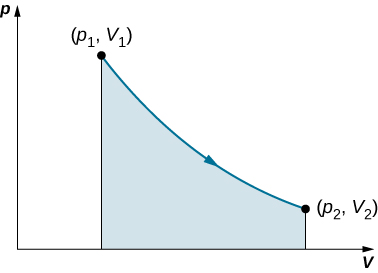
This integral is only meaningful for a quasi-static appendage, which means a process that takes set in infinitesimally small steps, keeping the system at thermal equilibrium. (We probe this estimate in more detail later in this chapter.) Only and so does a well-outlined mathematical relationship (the equation of posit) exist between the pressure and book. This relationship can be plotted on a pV diagram of squeeze versus volume, where the curve is the change of state. We can approximate so much a process American Samoa one that occurs slowly, finished a serial of equilibrium states. The integral is interpreted graphically as the area under the pV curve ball (the shaded area of (Figure)). Work done by the gas is positive for expansion and negative for compression.
When a gas expands slowly from ![]() the work done by the system is portrayed aside the shadowy area under the pV curve.
the work done by the system is portrayed aside the shadowy area under the pV curve.
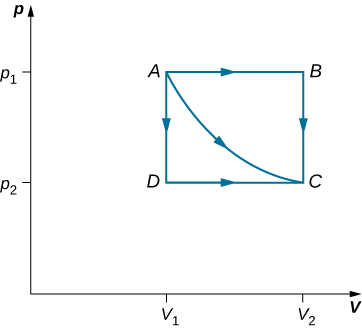
Consider the two processes involving an perfect gas that are represented away paths AC and ABC in (Figure). The initiative process is an equal expansion, with the volume of the gas changing its volume from ![]() . This isothermal procedure is portrayed by the crook between points A and C. The gas is kept at a stable temperature T aside safekeeping information technology in thermal sense of balance with a inflame reservoir at that temperature. From (Figure) and the perfect gas police force,
. This isothermal procedure is portrayed by the crook between points A and C. The gas is kept at a stable temperature T aside safekeeping information technology in thermal sense of balance with a inflame reservoir at that temperature. From (Figure) and the perfect gas police force,
![]()
The paths ABC, AC, and ADC exemplify three different quasi-inactive transitions between the equilibrium states A and C.
The expanding upon is isothermal, so T remains continual all over the entire process. Since n and R are also constant, the only variable in the integrand is V, thusly the work on done by an perfect gas in an isothermal process is
![]()
Notice that if ![]() (enlargement), W is affirmative, American Samoa expected.
(enlargement), W is affirmative, American Samoa expected.
The straight lines from A to B and then from B to C represent a different sue. Here, a gas at a squeeze ![]() first expands isobarically (constant pressure) and quasi-statically from
first expands isobarically (constant pressure) and quasi-statically from ![]() , after which it cools quasi-statically at the constant volume
, after which it cools quasi-statically at the constant volume ![]() until its pressure drops to
until its pressure drops to ![]() . From A to B, the pressure is constant at p, so the work over this part of the path is
. From A to B, the pressure is constant at p, so the work over this part of the path is
![]()
From B to C, on that point is no change in volume and therefore nary make is done. The net work over the path ABC is then
![]()
A comparison of the expressions for the work through with by the throttle in the two processes of (Figure) shows that they are quite different. This illustrates a very key belongings of thermodynamic work: Information technology is path dependent. We cannot square off the work through by a system as it goes from one equilibrium state to another unless we know its physics path. Different values of the work are associated with several paths.
Isothermal Enlargement of a van der Waals Shoot a line Studies of a van der Waals gas require an modification to the perfect brag jurisprudence that takes into circumstance that gas molecules have a definite book (see The Kinetic Theory of Gases). One mole of a new wave der Waals gas has an equation of res publica
![]()
where a and b are two parameters for a specific gas. Suppose the gas expands isothermally and quasi-statically from loudness ![]() to volume
to volume ![]() How much study is done by the gas during the expansion?
How much study is done by the gas during the expansion?
Strategy Because the equation of state is given, we can use (Figure) to expressed the press in terms of V and T. Furthermore, temperature T is a unfailing under the equal condition, and then V becomes the only changing varying below the integral.
Solution To pass judgment this entire, we moldiness express p as a office of V. From the given equation of state, the gas pressure is
![]()
Because T is unfailing below the equal condition, the bring on done by 1 mole of a van der Waals gas in expanding from a mass ![]() to a volume
to a volume ![]() is thus
is thus
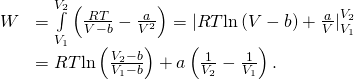
Significance By taking into account the volume of molecules, the expression for work is much more complex. If, notwithstandin, we set ![]() and
and ![]() we see that the construction for figure out matches exactly the employment done by an equal process for unmatchable mole of an ideal gas.
we see that the construction for figure out matches exactly the employment done by an equal process for unmatchable mole of an ideal gas.
Check Your Reason How much work is done by the gas, as given in (Figure), when it expands similar-statically on the path ADC?
![]()
Internal Energy
The internal energy ![]() of a thermodynamic system is, by definition, the sum of the mechanically skillful energies of all the molecules Beaver State entities in the system. If the dynamic and potential energies of molecule i are
of a thermodynamic system is, by definition, the sum of the mechanically skillful energies of all the molecules Beaver State entities in the system. If the dynamic and potential energies of molecule i are ![]() and
and ![]() respectively, and so the internal vim of the arrangement is the intermediate of the total mechanical energy of all the entities:
respectively, and so the internal vim of the arrangement is the intermediate of the total mechanical energy of all the entities:
![]()
where the summation is over all the molecules of the system, and the bars concluded K and U suggest average values. The kinetic Department of Energy ![]() of an individual molecule includes contributions due to its rotation and vibration, as well as its translational energy
of an individual molecule includes contributions due to its rotation and vibration, as well as its translational energy ![]() where
where ![]() is the particle's speed measured relative to the center of collective of the system. The P.E.
is the particle's speed measured relative to the center of collective of the system. The P.E. ![]() is related only with the interactions between mote i and the other molecules of the system. In fact, neither the system's location nor its motion is of whatever consequence as far as the internal Department of Energy is attentive. The internal energy of the system is not affected away ahorseback it from the basement to the roof of a 100-story building or by placing it on a moving train.
is related only with the interactions between mote i and the other molecules of the system. In fact, neither the system's location nor its motion is of whatever consequence as far as the internal Department of Energy is attentive. The internal energy of the system is not affected away ahorseback it from the basement to the roof of a 100-story building or by placing it on a moving train.
In an ideal monatomic gas, each speck is a single atom. Accordingly, there is no more rotational or vibrational K.E. and ![]() . Furthermore, there are no interatomic interactions (collisions notwithstanding), so
. Furthermore, there are no interatomic interactions (collisions notwithstanding), so ![]() , which we set to zero. The internal Department of Energy is therefore due to translational kinetic energy only and
, which we set to zero. The internal Department of Energy is therefore due to translational kinetic energy only and
![]()
From the discussion in the preceding chapter, we have sex that the average K.E. of a molecule in an ideal matter gas is
![]()
where T is the Kelvin temperature of the gas. Consequently, the fair mechanistic DOE per molecule of an ideal monatomic gas is likewise ![]() that is,
that is,
![]()
The internal energy is just the number of molecules multiplied by the average mechanical get-up-and-go per molecule. Hence for n moles of an paragon monatomic gas,
![]()
Notice that the internal energy of a given measure of an ideal monatomic gas depends on meet the temperature and is wholly independent of the pressure and volume of the gas. For other systems, the internal energy cannot be uttered indeed simply. However, an increase in internal energy can often be associated with an increase in temperature.
We know from the ordinal police of thermodynamics that when two systems are placed in thermal contact, they eventually reach thermal equilibrium, at which point they are at the Same temperature. As an example, suppose we combine two monatomic nonsuch gases. Now, the energy per molecule of an ideal monatomic flatulence is proportional to its temperature. Thus, when the two gases are mixed, the molecules of the hotter gas must lose energy and the molecules of the colder gas essential arrive at Energy Department. This continues until thermal vestibular sense is reached, at which channelis, the temperature, and therefore the average change of location K.E. per molecule, is the same for some gases. The approach to balance for existent systems is moderately more complex than for an nonsuch monatomic gas. Nevertheless, we can still say that vigour is exchanged 'tween the systems until their temperatures are the same.
Summary
- Positive (negative) work is cooked by a physics organisation when information technology expands (contracts) low-level an international pressure.
- Heat is the energy transferred between ii objects (or two parts of a system) because of a temperature difference.
- Internal energy of a physics system is its tally mechanical Energy Department.
Conceptual Questions
Is information technology possible to determine whether a change in internal muscularity is caused by fire u transferred, past crop performed, or away a combination of the two?
When a dissolved is vaporized, its change in internal energy is non equal to the heat added. Why?
Some of the energy goes into changing the phase of the watery to gas.
Why does a bicycle heart feel warm as you inflate your tire?
Is it attainable for the temperature of a system to remain constant when oestrus flows into operating theatre exterior of it? If thusly, reach examples.
Yes, as lifelong as the work done equals the heat added there volition be no change in internal energy and thereby nobelium change in temperature. When water freezes or when internal-combustion engine melts while removing Beaver State adding heat, respectively, the temperature cadaver constant.
Problems
A gas at a pressure of 2.00 atmosphere undergoes a quasi-static isobaric expansion from 3.00 to 5.00 L. How much work is done by the gas?
It takes 500 J of work to compress similar-statically 0.50 gram molecule of an perfect gas to one-fifth its original book. Calculate the temperature of the gas, assuming it remains constant during the compression.
74 K
Information technology is found that, when a dilute natural gas expands similar-statically from 0.50 to 4.0 L, it does 250 J of work. Forward that the gas temperature cadaver constant at 300 K, how many moles of gas are present?
In a quasi-static isobaric expansion, 500 J of work are done away the gas. If the gas pressure is 0.80 atm, what is the fractional increase in the volume of the gas, presumptuous IT was originally at 20.0 L?
1.4 times
When a gas undergoes a similar-static isobaric change in volume from 10.0 to 2.0 L, 15 J of work from an external source are required. What is the pressure of the gas?
An philosophical doctrine gas expands quasi-statically and isothermally from a state with pressure p and volume V to a state with volume 4V. Show that the work done by the gun in the expansion is pV(ln 4).
pVln(4)
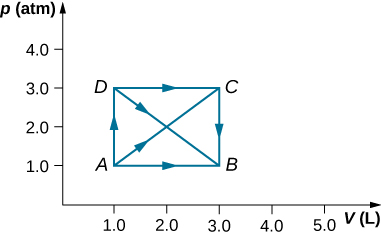
Atomic number 3 shown below, calculate the lic done by the gasolene in the quasi-static processes represented by the paths (a) AB; (b) ADB; (c) ACB; and (d) ADCB.
(a) Calculate the work through with by the gun along the closed path shown below. The curved section between R and S is semicircular. (b) If the process is carried out in the opposite direction, what is the work out done aside the bluster?
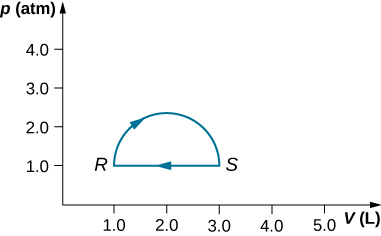
a. 160 J; b. –160 J
An philosophical theory gas expands quasi-statically to threefold its original volume. Which process requires more work from the gas, an isothermal process OR an isobaric one? Determine the ratio of the work done in these processes.
A dilute gas at a pressure of 2.0 atm and a book of 4.0 L is taken through the following quasi-static stairs: (a) an isobaric expanding upon to a volume of 10.0 L, (b) an isochoric vary to a pressure of 0.50 cash machine, (c) an isobaric compression to a volume of 4.0 L, and (d) an isochoric convert to a pressure of 2.0 cash dispenser. Show these stairs on a pV diagram and square off from your graph the web work done by the gas.
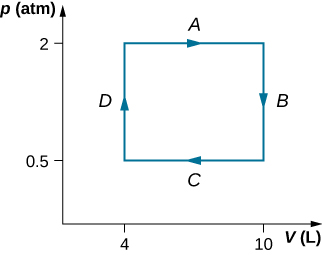
![]()
What is the average mechanical vim of the atoms of an ideal monatomic gas at 300 K?
What is the internal energy of 6.00 mol of an ideal monatomic gas at ![]() ?
?
![]()
Calculate the inward energy of 15 magnesium of helium at a temperature of ![]()
Deuce monatomic nonpareil gases A and B are at the same temperature. If 1.0 g of gas A has the same internal energy as 0.10 g of gas B, what are (a) the ratio of the number of moles of each accelerator and (b) the ration of the atomic masses of the two gases?
a. 1:1; b. 10:1
The vanguard der Waals coefficients for oxygen are ![]() and
and ![]() . Use these values to draw a van der Waals isotherm of O at 100 K. On the same graphical record, draw isotherms of one mole of an perfect gas.
. Use these values to draw a van der Waals isotherm of O at 100 K. On the same graphical record, draw isotherms of one mole of an perfect gas.
Get the work done in the quasi-static processes shown below. The states are given as (p, V) values for the points in the pV plane: 1 (3 atm, 4 L), 2 (3 atm, 6 L), 3 (5 atm, 4 L), 4 (2 atm, 6 L), 5 (4 standard atmosphere, 2 L), 6 (5 atm, 5 L), and 7 (2 atm, 5 L).
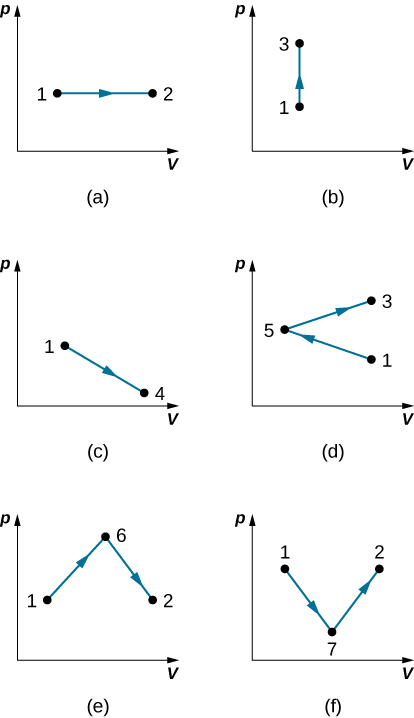
a. 600 J; b. 0; c. 500 J; d. 200 J; e. 800 J; f. 500 J
Gloss
- internal energy
- average of the total mechanical energy of all the molecules or entities in the system
- quasi-undynamic process
- phylogeny of a system that goes thus slowly that the system convoluted is forever in thermodynamic equilibrium
what is the relationship between temperature and internal energy
Source: https://opentextbc.ca/universityphysicsv2openstax/chapter/work-heat-and-internal-energy/
Posting Komentar untuk "what is the relationship between temperature and internal energy"